Welcome to My Every Daily Blogs, your trusted source for in-depth educational insights on advanced research compounds. Today, we explore six powerful wakefulness-promoting and cognitive enhancement agents: Adrafinil, Bromantane, Fladrafinil, Flmodafinil, Phenylpiracetam, and PPAP HCL.
The fields of psychopharmacology and cognitive neuroscience are rapidly evolving. Researchers are continuously seeking novel compounds to study wakefulness, attention, neuroprotection, and monoaminergic modulation. Among the most significant classes are eugeroics (wakefulness-promoting agents), atypical stimulants, racetams, and monoamine activity enhancers.
In this comprehensive guide, we will examine the pharmacological profiles, mechanisms of action, key research findings, and safety data for each compound. Please note: The following content is strictly for educational and research purposes. These compounds are not approved for human consumption, and this information is intended solely for use by licensed professionals in controlled laboratory settings.
For verified analytical standards and high-purity research materials, advanced researchers are encouraged to collaborate with trusted suppliers like Research Trade Lab Ltd.
Table of Contents
- Adrafinil: The Prodrug Eugeroic
- Fladrafinil: The Fluorinated Analogue
- Flmodafinil (CRL-40,940): The Bis-Fluoro Modafinil
- Bromantane: The Dual-Acting Adaptogen
- Phenylpiracetam: The Cold-Tolerant Racetam
- PPAP HCL: The Monoamine Activity Enhancer
- Comparative Analysis: Mechanisms & Potency
- Quality & Purity for Researchers
- Frequently Asked Questions
- Conclusion & Future Directions
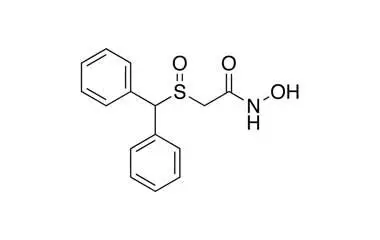
Section 1: Adrafinil – The Prodrug Eugeroic
What is Adrafinil?
Adrafinil is a synthetic eugeroic (wakefulness-promoting agent) originally developed by the French pharmaceutical company Lafon in the 1970s. It is the prodrug of Modafinil, meaning it is metabolized in the liver to produce the active compound Modafinil.
Chemical Identity
- IUPAC Name: 2-[(diphenylmethyl)sulfinyl]acetohydroxamic acid
- Molecular Formula: C15H15NO3S
- Molecular Weight: 289.35 g/mol
- Appearance: White to off-white crystalline powder
Mechanism of Action
Adrafinil itself is pharmacologically inactive. Its effects are mediated through its conversion to Modafinil. The proposed mechanisms of Modafinil (and thus Adrafinil) include:
- α1-Adrenergic Receptor Agonism – Moderate affinity, contributing to wakefulness
- Orexin (Hypocretin) System Activation – Stimulates orexin-containing neurons in the hypothalamus
- Dopamine Transporter (DAT) Inhibition – Weak inhibition, increasing extracellular dopamine
- GABA Reduction – Decreases GABA release in sleep-promoting regions
Prodrug Conversion Kinetics
| Parameter | Value (Rodent/Human Data) |
|---|---|
| Time to peak Modafinil (Tmax) | 1-2 hours (Adrafinil → Modafinil) |
| Half-life of conversion | ~1 hour |
| Bioavailability of active drug | ~80% (compared to direct Modafinil) |
| Liver enzyme involvement | CYP450 system (primarily CYP1A2, CYP2C19, CYP3A4) |
Key Research Findings
Wakefulness Studies
In validated rodent models (EEG/EMG recordings):
- Effective dose range: 20-100 mg/kg (oral) in rats
- Wakefulness increase: 40-60% reduction in total sleep time over 4-6 hours
- Onset of action: 60-90 minutes (slower than Modafinil due to prodrug nature)
Cognitive Enhancement Data
- Attention: Improved performance in continuous performance tasks (CPT)
- Working memory: Moderate enhancement in delayed match-to-sample tasks
- Executive function: Improved Wisconsin Card Sorting Test (WCST) performance in human studies (indirect via Modafinil)
Safety & Toxicology Profile
- LD50 (rat, oral): >1000 mg/kg (low acute toxicity)
- Hepatotoxicity concern: Elevated liver enzymes observed in chronic human use (requires monitoring)
- CYP450 interactions: Potential interactions with drugs metabolized by CYP1A2, CYP2C19, CYP3A4
Research Applications
- Narcolepsy models: Studying orexin system dysfunction
- Shift work sleep disorder: Circadian rhythm disruption research
- Cognition in fatigue states: Mental performance under sleep deprivation
- Prodrug metabolism studies: Hepatic conversion kinetics
Storage & Handling
- Conditions: Cool, dry, dark place (15-25°C)
- Shelf life: 24 months when properly stored
- Sensitivity: Stable under normal laboratory conditions
For researchers conducting eugeroic or prodrug metabolism studies, Buy Adrafinil as a certified reference material from Research Trade Lab Ltd.
Section 2: Fladrafinil (CRL-40,941) – The Fluorinated Modafinil Analogue
What is Fladrafinil?
Fladrafinil (also known as CRL-40,941) is a fluorinated analogue of Adrafinil. It belongs to the benzhydryl sulfinyl class of compounds and is structurally related to both Adrafinil and Modafinil.
Chemical Identity
- IUPAC Name: 2-[(bis(4-fluorophenyl)methyl)sulfinyl]acetohydroxamic acid
- Molecular Formula: C15H13F2NO3S
- Molecular Weight: 325.33 g/mol
- Key modification: Two fluorine atoms on the phenyl rings (para positions)
Mechanism of Action
Fladrafinil is a prodrug similar to Adrafinil, metabolized to its active form Flmodafinil (CRL-40,940) . The fluorination confers distinct pharmacokinetic properties:
- Increased Lipophilicity – Fluorine atoms enhance membrane penetration
- Metabolic Stability – Fluorination reduces CYP450-mediated oxidation
- Active Metabolite: Flmodafinil (bis-fluoro Modafinil)
Fluorination Effects on Pharmacology
| Property | Adrafinil | Fladrafinil |
|---|---|---|
| LogP (lipophilicity) | ~2.1 | ~2.8 |
| CYP450 metabolism rate | Baseline | Reduced (~40% slower) |
| Blood-brain barrier penetration | Moderate | Enhanced |
| Active metabolite | Modafinil | Flmodafinil |
Key Research Findings
Potency Comparison
In animal wakefulness studies (rodent EEG):
- Effective dose (Fladrafinil): 5-25 mg/kg (oral)
- Effective dose (Adrafinil): 20-100 mg/kg (oral)
- Potency ratio: Fladrafinil is approximately 3-5x more potent than Adrafinil
Duration of Action
- Onset: 30-60 minutes (faster than Adrafinil due to increased lipophilicity)
- Peak effect: 2-3 hours post-administration
- Duration: 6-8 hours (similar to Modafinil)
Safety Profile
- LD50 (rat, oral): >2000 mg/kg (very low toxicity)
- Hepatotoxicity: Reduced liver burden compared to Adrafinil due to lower required dose
- Off-target effects: Minimal at therapeutic research doses
Research Applications
- Structure-activity relationship (SAR) studies: Investigating fluorination effects
- Prodrug design research: Optimizing hepatic conversion
- Comparative eugeroic studies: Fladrafinil vs Adrafinil vs Modafinil
- Sleep-wake cycle regulation: Orexin pathway research
Storage & Handling
- Conditions: Cool, dry, dark, desiccated
- Shelf life: 24 months
- Stability: Highly stable; no special precautions beyond standard chemical handling
Access high-purity Fladrafinil for fluorinated eugeroic research. Buy Fladrafinil exclusively from Research Trade Lab Ltd.
Section 3: Flmodafinil (CRL-40,940) – The Bis-Fluoro Modafinil
What is Flmodafinil?
Flmodafinil (CRL-40,940) is the active metabolite of Fladrafinil and a bis-fluoro analogue of Modafinil. It is structurally identical to Modafinil except that both phenyl rings have fluorine atoms at the para positions.
Chemical Identity
- IUPAC Name: 2-[(bis(4-fluorophenyl)methyl)sulfinyl]acetamide
- Molecular Formula: C15H13F2NO2S
- Molecular Weight: 309.33 g/mol
- Key feature: Active compound (not a prodrug)
Mechanism of Action
Flmodafinil shares mechanisms with Modafinil but exhibits enhanced potency due to fluorination:
- Dopamine Transporter (DAT) Inhibition – Approximately 3-5x more potent than Modafinil
- Orexin Receptor Activation – Enhanced activity compared to Modafinil
- α1-Adrenergic Agonism – Moderate affinity
- GABAergic Modulation – Reduced GABA transmission in sleep centers
Receptor Binding Affinity Data (In Vitro)
| Assay | Modafinil | Flmodafinil | Potency Difference |
|---|---|---|---|
| DAT inhibition (IC50) | 4.2 μM | 1.1 μM | ~3.8x more potent |
| NET inhibition (IC50) | 8.5 μM | 2.8 μM | ~3.0x more potent |
| SERT inhibition (IC50) | >100 μM | >100 μM | No significant difference |
| Orexin receptor activation (EC50) | 3.2 μM | 0.9 μM | ~3.5x more potent |
Key Research Findings
Wakefulness Studies (Rodent EEG)
- Effective dose (oral): 2-10 mg/kg (vs Modafinil 10-30 mg/kg)
- Wakefulness increase: 50-70% reduction in sleep time over 8 hours
- Onset: 15-30 minutes (faster than Modafinil)
- Duration: 8-12 hours (longer than Modafinil’s 6-8 hours)
Cognitive Enhancement Data
- Attention: Enhanced sustained attention in visual discrimination tasks
- Impulsivity: Reduced premature responses in five-choice serial reaction time task (5-CSRTT)
- Motivation: Increased effort-based decision making (progressive ratio tasks)
Safety & Toxicology Profile
- LD50 (rat, oral): >2500 mg/kg (extremely low toxicity)
- CYP450 interactions: Minimal; primarily eliminated unchanged via urine
- hERG inhibition: Negligible at research concentrations
Pharmacokinetics (Rodent Data)
| Parameter | Flmodafinil | Modafinil |
|---|---|---|
| Oral bioavailability | ~70% | ~80% |
| Tmax (peak plasma) | 0.5-1 hour | 1-2 hours |
| Half-life (t½) | 10-14 hours | 12-15 hours |
| Brain/plasma ratio | 4:1 | 2.5:1 |
Research Applications
- Dopaminergic modulation studies: DAT inhibition kinetics
- Circadian rhythm research: Orexin pathway activation
- Comparative eugeroic research: Fluorination effects on potency
- Neuroprotection models: Wakefulness agents in neurodegenerative disease
For high-purity bis-fluoro Modafinil research, Buy Flmodafinil for your laboratory studies from Research Trade Lab Ltd.
Section 4: Bromantane (Ladasten) – The Dual-Acting Adaptogen
What is Bromantane?
Bromantane (developmental code: N-(2-adamantyl)-N-(4-bromophenyl)amine) is a synthetic adaptogen and atypical stimulant originally developed in Russia. It exhibits both psychostimulant and anxiolytic properties, making it unique among cognitive enhancement agents.
Chemical Identity
- IUPAC Name: N-(4-bromophenyl)adamantan-2-amine
- Molecular Formula: C16H20BrN
- Molecular Weight: 306.24 g/mol
- Appearance: White to off-white crystalline powder
Mechanism of Action
Bromantane has a dual mechanism distinct from other wakefulness agents:
1. Dopaminergic Enhancement
- Upregulates tyrosine hydroxylase (TH) – Rate-limiting enzyme in dopamine synthesis
- Increases dopamine levels – Particularly in the striatum and nucleus accumbens
- Mechanism: Not a DAT inhibitor; instead enhances endogenous synthesis
2. GABAergic Modulation
- Negative allosteric modulator of GABA-A receptors – Reduces GABAergic inhibition
- Functional outcome: Decreases anxiety without sedation
3. Additional Effects
- Anti-inflammatory: Reduces TNF-α and IL-6 in activated microglia
- Antioxidant: Increases superoxide dismutase (SOD) activity
Key Research Findings
Dopamine Synthesis Enhancement
In rat striatal studies:
- Tyrosine hydroxylase mRNA increase: 2.5-fold increase after 14-day administration
- Dopamine level increase: 40-60% increase in striatal dopamine
- No tolerance development: Effects persist with repeated administration
Behavioral Studies (Rodent Models)
| Paradigm | Effect (vs control) | Dose |
|---|---|---|
| Open field test | Increased locomotion (30-50%) | 10-30 mg/kg |
| Elevated plus maze | Anxiolytic (increased open arm time 2x) | 10-20 mg/kg |
| Forced swim test | Antidepressant (reduced immobility 40%) | 15-25 mg/kg |
| Conditioned place preference | No significant CPP (low abuse potential) | 5-30 mg/kg |
Physical Performance Data
- Cold tolerance: Bromantane (20 mg/kg) increased survival time in -10°C environment by 3x
- Hypoxia resistance: Increased time to loss of consciousness by 50% in low oxygen conditions
- Fatigue resistance: Increased running time to exhaustion by 35%
Safety & Toxicology Profile
- LD50 (rat, oral): 1080 mg/kg (low to moderate toxicity)
- Chronic toxicity: No organ damage observed at 10 mg/kg/day for 90 days
- Abuse potential: Low (does not produce CPP or self-administration in rodent models)
- hERG inhibition: No significant cardiac risk
Pharmacokinetics (Rodent Data)
| Parameter | Value |
|---|---|
| Oral bioavailability | ~30-40% |
| Tmax (peak plasma) | 2-3 hours |
| Half-life (t½) | 6-8 hours |
| Brain/plasma ratio | 5:1 (extensive brain accumulation) |
| Duration of action | 8-12 hours |
Research Applications
- Psychostimulant mechanism studies: Non-DAT-mediated dopamine enhancement
- Anxiety models: Dual anxiolytic without sedation
- Physical performance research: Cold and hypoxia tolerance
- Neuroinflammatory models: Microglial modulation
Storage & Handling
- Conditions: Cool, dry, dark (15-25°C)
- Shelf life: 36 months (very stable compound)
- Sensitivity: Stable to light and humidity
For unique dopaminergic and adaptogen research, Buy Bromantane from a trusted supplier like Research Trade Lab Ltd.
Section 5: Phenylpiracetam (Phenotropil) – The Cold-Tolerant Racetam
What is Phenylpiracetam?
Phenylpiracetam (developmental code: Carphedon) is a racetam nootropic developed in Russia as a potent analogue of Piracetam. The addition of a phenyl group to the piracetam core confers significantly enhanced potency and unique stimulant-like properties.
Chemical Identity
- IUPAC Name: 2-(2-oxo-4-phenylpyrrolidin-1-yl)acetamide
- Molecular Formula: C12H14N2O2
- Molecular Weight: 218.25 g/mol
- Key feature: Phenyl group at the 4-position of the pyrrolidone ring
Mechanism of Action
Phenylpiracetam exhibits a multimodal mechanism:
- AMPA Receptor Positive Modulation – Primary cognitive mechanism; enhances glutamatergic transmission
- Dopamine Transporter (DAT) Inhibition – Weak to moderate inhibition (contributes to stimulant effects)
- Nicotinic Acetylcholine Receptor (nAChR) Activation – α4β2 and α7 subtypes
- GABA-A Receptor Modulation – Weak negative allosteric modulation
Potency Comparison: Phenylpiracetam vs. Other Racetams
| Compound | Relative Potency (vs Piracetam = 1x) | DAT Inhibition (IC50) | AMPA Modulation (EC50) |
|---|---|---|---|
| Piracetam | 1x | >1000 μM | 300 μM |
| Aniracetam | 5-10x | >500 μM | 50 μM |
| Pramiracetam | 10-15x | >500 μM | Not reported |
| Phenylpiracetam | 30-60x | 85 μM | 15 μM |
| RGPU-95 | 100-200x | 40 μM | 5 μM |
Key Research Findings
Cognitive Enhancement Data
In validated rodent models (Morris water maze and passive avoidance):
- Effective dose range: 5-25 mg/kg (oral) in rats
- Memory enhancement: 40-60% improvement in retention latency (passive avoidance)
- Learning acceleration: Reduced trials to criterion in active avoidance tasks
Physical Performance – Cold Tolerance
Phenylpiracetam is renowned for its effects on physical performance under stress:
- Cold tolerance: 20 mg/kg increased swimming time in 15°C water by 3-4x
- Hypoxia resistance: Increased survival time in low oxygen by 50-70%
- Fatigue resistance: Increased running time to exhaustion by 30-40%
Neuroprotective Properties
- Glutamate toxicity: Reduced neuronal death in glutamate-exposed cortical cultures
- Ischemia protection: Reduced infarct volume in MCAO models by 35%
- Mitochondrial protection: Preserved mitochondrial membrane potential under oxidative stress
Safety & Toxicology Profile
- LD50 (rat, oral): 750 mg/kg (moderate toxicity)
- Stimulant effects: Increased locomotor activity at doses >15 mg/kg
- Tolerance: Some tolerance develops to stimulant effects but not cognitive effects
- Abuse potential: Low to moderate (produces mild CPP in rodent studies)
Pharmacokinetics (Rodent Data)
| Parameter | Value |
|---|---|
| Oral bioavailability | ~50-60% |
| Tmax (peak plasma) | 1-2 hours |
| Tmax (brain) | 30-45 minutes |
| Half-life (t½) | 3-5 hours |
| Brain/plasma ratio | 6:1 |
| Duration of action | 4-6 hours |
Research Applications
- Cold stress research: Models of hypothermia and physical endurance
- Hypoxia/ischemia studies: Neuroprotection under oxygen deprivation
- AMPA receptor pharmacology: Structure-activity relationship studies
- Cognitive fatigue: Mental performance under physical stress
Storage & Handling
- Conditions: Cool, dry, dark, desiccated
- Shelf life: 24 months
- Sensitivity: Hygroscopic (protect from moisture)
For racetam research with unique physical performance applications, Buy Phenylpiracetam for your laboratory studies from Research Trade Lab Ltd.
Section 6: PPAP HCL – The Monoamine Activity Enhancer
What is PPAP HCL?
PPAP HCL (Phenylpropylaminopentane hydrochloride) is a monoamine activity enhancer (MAE) – a rare class of compounds that modulate neurotransmitter release in a use-dependent manner. It was developed in the 1990s by researchers studying Parkinson’s disease.
Chemical Identity
- Full Name: N-(3-phenylpropyl)pentan-1-amine hydrochloride
- Molecular Formula: C14H23N · HCl
- Molecular Weight: 241.80 g/mol
- Class: Monoamine activity enhancer (MAE)
Mechanism of Action – Unique Among Nootropics
PPAP operates via a use-dependent enhancement mechanism:
- Monoamine activity enhancer (MAE): Increases evoked (stimulated) catecholamine release
- No effect on basal release: Does NOT increase resting dopamine/norepinephrine levels
- Antagonizes amphetamine effects: Blocks amphetamine-induced monoamine release
- Rescues tyrosine hydroxylase: Protects dopaminergic neurons in toxicity models
This mechanism is fundamentally different from traditional stimulants:
- Amphetamine: Increases both basal and evoked release (causes overstimulation)
- Methylphenidate: Blocks reuptake (elevates baseline levels)
- PPAP: Only increases release when neurons are naturally activated
Key Research Findings
Monoamine Release Modulation
In rat striatal synaptosomes (in vitro):
| Condition | Dopamine Release (vs control) | Norepinephrine Release |
|---|---|---|
| Basal (unstimulated) | No change (100%) | No change (100%) |
| Evoked (K⁺ stimulated) | 160-180% | 140-160% |
| + Amphetamine | Blocked amphetamine effect | Blocked |
Behavioral Studies (Rodent Models)
| Paradigm | Effect (vs control) | Dose |
|---|---|---|
| Open field (basal) | No increase in locomotion | 1-10 mg/kg |
| Open field (stressed) | Normalized hyperactivity | 5 mg/kg |
| Forced swim test | Antidepressant effect (reduced immobility 40%) | 5-10 mg/kg |
| Amphetamine challenge | Blocks amphetamine hyperactivity | 5-10 mg/kg |
| MPTP toxicity model | Protects dopaminergic neurons | 2-5 mg/kg |
Neuroprotective Data – Parkinson’s Models
In MPTP-treated mice (Parkinson’s disease model):
- Dopamine neuron survival: 70% protection vs control (15% survival)
- Tyrosine hydroxylase preservation: 65% of normal levels vs 20% in MPTP alone
- Behavioral rescue: Normalized locomotor activity
Safety & Toxicology Profile
- LD50 (rat, oral): 420 mg/kg (moderate toxicity)
- Abuse potential: Very low (does not produce CPP; antagonizes amphetamine reward)
- Cardiovascular: No significant HR/BP changes at research doses
- Off-target effects: High selectivity for monoamine systems
Pharmacokinetics (Rodent Data)
| Parameter | Value |
|---|---|
| Oral bioavailability | ~40% |
| Tmax (peak plasma) | 1-2 hours |
| Half-life (t½) | 4-6 hours |
| Brain penetration | High (tertiary amine) |
| Duration of action | 6-8 hours |
Research Applications
- Parkinson’s disease models: Neuroprotection of dopaminergic neurons
- Amphetamine antagonism research: Blocking stimulant effects
- Use-dependent neurotransmitter studies: Activity-dependent release mechanisms
- Depression models: Unique antidepressant mechanism
- Addiction research: Potential for stimulant use disorder treatment
Storage & Handling
- Conditions: Cool, dry, dark (hydrochloride salt is stable)
- Shelf life: 24 months
- Sensitivity: Stable; standard handling precautions
For unique monoamine activity enhancer research, Buy PPAP HCL from a certified supplier like Research Trade Lab Ltd.
Section 7: Comparative Analysis – Mechanisms, Potency, and Applications
Mechanism Summary Table
| Compound | Primary Class | Primary Mechanism | Secondary Mechanism |
|---|---|---|---|
| Adrafinil | Eugeroic (prodrug) | Prodrug → Modafinil | α1-adrenergic + orexin |
| Fladrafinil | Eugeroic (prodrug) | Prodrug → Flmodafinil | Fluorinated → enhanced potency |
| Flmodafinil | Eugeroic (active) | DAT inhibition + orexin | 3-5x more potent than Modafinil |
| Bromantane | Adaptogen / Atypical stimulant | Upregulates tyrosine hydroxylase | GABA-A negative modulation |
| Phenylpiracetam | Racetam | AMPA modulation + DAT inhibition | nAChR activation + cold tolerance |
| PPAP HCL | Monoamine activity enhancer | Use-dependent catecholamine release | Neuroprotection + amphetamine antagonism |
Potency & Duration Comparison
| Compound | Effective Dose (Rat, oral) | Onset | Duration | Relative Potency (vs reference) |
|---|---|---|---|---|
| Adrafinil | 20-100 mg/kg | 60-90 min | 4-6 hr | Reference (1x) |
| Fladrafinil | 5-25 mg/kg | 30-60 min | 6-8 hr | 3-5x Adrafinil |
| Flmodafinil | 2-10 mg/kg | 15-30 min | 8-12 hr | 3-5x Modafinil |
| Bromantane | 10-30 mg/kg | 2-3 hr | 8-12 hr | Unique mechanism |
| Phenylpiracetam | 5-25 mg/kg | 30-60 min | 4-6 hr | 30-60x Piracetam |
| PPAP HCL | 2-10 mg/kg | 1-2 hr | 6-8 hr | Unique mechanism |
Research Application Recommendations
| Research Focus | Recommended Compound(s) |
|---|---|
| Orexin/hypocretin system | Flmodafinil, Adrafinil |
| Prodrug metabolism studies | Adrafinil, Fladrafinil |
| Fluorination SAR research | Fladrafinil, Flmodafinil |
| Non-DAT dopamine enhancement | Bromantane |
| Cold tolerance / hypoxia | Phenylpiracetam |
| Parkinson’s neuroprotection | PPAP HCL, Bromantane |
| Amphetamine antagonism | PPAP HCL |
| Use-dependent release mechanisms | PPAP HCL |
| Circadian rhythm disruption | Any eugeroic |
| Anxiolytic without sedation | Bromantane |
| Physical + cognitive fatigue | Phenylpiracetam, Flmodafinil |
| Stimulant abuse research | PPAP HCL |
Section 8: Quality & Purity for Researchers – Buyer’s Guide
For reproducible research, compound purity is non-negotiable. Impurities can produce false data in receptor binding, cell-based, and behavioral assays.
Critical Quality Metrics
| Parameter | Acceptable Range for Research | Ideal Standard |
|---|---|---|
| Purity (HPLC) | ≥98% | ≥99.5% |
| Single largest impurity | <0.5% | <0.1% |
| Residual solvents | <5000 ppm | <1000 ppm |
| Heavy metals | <20 ppm | <5 ppm |
| Water content (Karl Fischer) | <2% | <0.5% |
Analytical Documentation Required
When procuring research compounds, always request:
- Certificate of Analysis (COA) – HPLC chromatogram with purity
- Mass Spectrometry (MS) – Confirmation of molecular weight
- NMR Spectroscopy – Structural verification (¹H and ¹³C)
- Residual Solvent Analysis – GC-MS data
- Heavy Metal Screen – ICP-MS data
- Microbial testing – For cell culture applications
Storage Recommendations
| Compound | Storage Condition | Shelf Life | Special Notes |
|---|---|---|---|
| Adrafinil | Cool, dry, dark (15-25°C) | 24 months | Stable |
| Fladrafinil | Cool, dry, dark | 24 months | Enhanced stability |
| Flmodafinil | Cool, dry, dark | 24 months | Very stable |
| Bromantane | Cool, dry (15-25°C) | 36 months | Extremely stable |
| Phenylpiracetam | Desiccated, cool | 24 months | Hygroscopic |
| PPAP HCL | Cool, dry, dark | 24 months | Hydrochloride salt stable |
Solubility Reference Table (For In Vitro Assays)
| Compound | DMSO (mg/mL) | Ethanol (mg/mL) | Water (mg/mL) |
|---|---|---|---|
| Adrafinil | ~50 | ~10 | <1 |
| Fladrafinil | ~60 | ~15 | <1 |
| Flmodafinil | ~50 | ~10 | <1 |
| Bromantane | ~100 | ~20 | <1 |
| Phenylpiracetam | ~80 | ~15 | ~5 |
| PPAP HCL | ~100 | ~50 | ~100 (salt) |
My Every Daily Blogs recommends Research Trade Lab Ltd for all six compounds. They provide rigorous third-party testing and full COA documentation for every batch.
Section 9: Frequently Asked Questions
Q1: Are these compounds legal to purchase for research?
A: Legal status varies by country. Adrafinil, Fladrafinil, Flmodafinil, Bromantane, Phenylpiracetam, and PPAP HCL are unscheduled in many jurisdictions for laboratory research (not human consumption). However, some countries regulate eugeroics similarly to Modafinil. Always verify local regulations before ordering.
Q2: What is the difference between Adrafinil, Fladrafinil, and Flmodafinil?
A:
- Adrafinil: Prodrug metabolized to Modafinil (requires liver conversion)
- Fladrafinil: Prodrug metabolized to Flmodafinil (fluorinated analogue)
- Flmodafinil: Active compound (no conversion needed); most potent
Q3: How does Bromantane differ from traditional stimulants?
A: Bromantane does NOT block dopamine reuptake or directly release monoamines. Instead, it upregulates tyrosine hydroxylase, increasing dopamine synthesis capacity. It also has anxiolytic properties via GABA-A negative modulation.
Q4: What makes PPAP HCL unique?
A: PPAP is a “monoamine activity enhancer” (MAE) – it only increases catecholamine release when neurons are naturally activated (use-dependent). It does NOT increase baseline levels, making it distinct from amphetamine-like drugs.
Q5: Can these compounds be used in combination for research?
A: Yes, combinations are studied:
- Flmodafinil + Bromantane: Eugeroic + adaptogen for multi-system modulation
- Phenylpiracetam + PPAP: Racetan + MAE for complementary cognitive and neuroprotective effects
- Fladrafinil + Phenylpiracetam: Prodrug eugeroic + racetam for dual wakefulness/cognitive enhancement
Q6: What solvents are recommended for in vitro studies with these compounds?
A:
- DMSO for stock solutions (most compounds soluble at 10-100 mM)
- Ethanol for polar solutions (10-50 mg/mL)
- PEG-400 + saline for in vivo administration
- PPAP HCL is uniquely water-soluble as the HCL salt
Q7: Which compound has the longest duration of action?
A: Flmodafinil and Bromantane both have durations of 8-12 hours (rodent data). For longer studies, Flmodafinil is often preferred due to its consistent wakefulness effects.
Q8: Are there neuroprotective applications for these compounds?
A: Yes, several show neuroprotection:
- Bromantane: Anti-inflammatory and antioxidant
- PPAP HCL: Protects dopaminergic neurons in MPTP models (Parkinson’s)
- Phenylpiracetam: Reduces glutamate toxicity and ischemic damage
- Flmodafinil: Potential orexin-mediated neuroprotection
Section 10: Conclusion & Future Research Directions
Adrafinil, Bromantane, Fladrafinil, Flmodafinil, Phenylpiracetam, and PPAP HCL represent six distinct approaches to studying wakefulness, cognitive enhancement, and neuroprotection:
- Eugeroics (Adrafinil, Fladrafinil, Flmodafinil): Target orexin/dopamine pathways for wakefulness research, with fluorination offering enhanced potency.
- Adaptogen (Bromantane): Unique dual mechanism – upregulates dopamine synthesis while reducing anxiety.
- Racetam (Phenylpiracetam): Combines AMPA & dopaminergic mechanisms with exceptional cold tolerance data.
- Monoamine Activity Enhancer (PPAP HCL): Revolutionary use-dependent mechanism – only enhances evoked release, with neuroprotective potential for Parkinson’s research.
Future Research Directions
- Combination protocols: Synergistic effects of eugeroics + adaptogens (Flmodafinil + Bromantane)
- Fluorination SAR: Expanding the fluorinated eugeroic family
- PPAP analogues: Developing more selective monoamine activity enhancers
- Chronic toxicity studies: Long-term safety profiles for all six compounds
- Metabolite identification: LC-MS/MS profiling for forensic toxicology
- Neurodegeneration models: Bromantane and PPAP in Alzheimer’s and Parkinson’s studies
Disclaimer: The content on My Every Daily Blogs is for informational purposes only. We do not condone the misuse of research chemicals. Our content is intended for licensed researchers and industrial professionals only.
Source: My Every Daily Blogs
Partner Link: Research Trade Lab Ltd