Welcome to My Every Daily Blogs, your trusted source for in-depth educational insights on advanced research compounds. Today, we explore eleven significant fluorinated stimulants and novel formulations: 2-FMA, 2-FEA, 2-FPM, 2/3-FEA, 3-FA, 3-FPM, 3-MMA, 3-FEA, 4-FMA, 4F-MPH, and Blue Mandala Mix.*
The field of stimulant pharmacology is rapidly advancing, with researchers continuously seeking novel compounds to study monoamine transporter interactions, structure-activity relationships (SAR), and positional isomerism effects. Fluorinated amphetamines, phenmetrazine analogues, and piperidine stimulants represent some of the most significant classes of research chemicals in this domain.
In this comprehensive guide, we will examine the pharmacological profiles, mechanisms of action, key research findings, and safety data for each compound. Please note: The following content is strictly for educational and research purposes. These compounds are not approved for human consumption, and this information is intended solely for use by licensed professionals in controlled laboratory settings.
For verified analytical standards and high-purity research materials, advanced researchers are encouraged to collaborate with trusted suppliers like Research Trade Lab Ltd.
Table of Contents
- Understanding Fluorinated Stimulants
- 2-FMA (2-Fluoromethamphetamine)
- 2-FEA (2-Fluoroethamphetamine)
- 2-FPM (2-Fluorophenmetrazine)
- 2/3-FEA (Positional Isomer Blend)
- 3-FA (3-Fluoroamphetamine)
- 3-FPM (3-Fluorophenmetrazine)
- 3-MMA (3-Methoxymethamphetamine)
- 3-FEA (3-Fluoroethamphetamine)
- 4-FMA (4-Fluoromethamphetamine)
- 4F-MPH (4-Fluoromethylphenidate)
- Blue Mandala Mix
- Positional Isomerism & SAR Comparison
- Quality & Purity for Researchers
- Frequently Asked Questions
- Conclusion & Future Directions
Section 1: Understanding Fluorinated Stimulants
What Are Fluorinated Stimulants?
Fluorinated stimulants are research chemicals belonging to the amphetamine, phenmetrazine, and phenidate classes, where one or more hydrogen atoms have been replaced with fluorine atoms. This fluorination significantly alters pharmacological properties including:
- Metabolic stability – Fluorine resists enzymatic degradation
- Lipophilicity – Enhanced blood-brain barrier penetration
- Receptor binding affinity – Altered selectivity for dopamine (DAT), norepinephrine (NET), and serotonin (SERT) transporters
Positional Isomerism
The position of the fluorine atom on the aromatic ring (2-position, 3-position, or 4-position) dramatically affects the pharmacological profile, making these compounds valuable for SAR research.
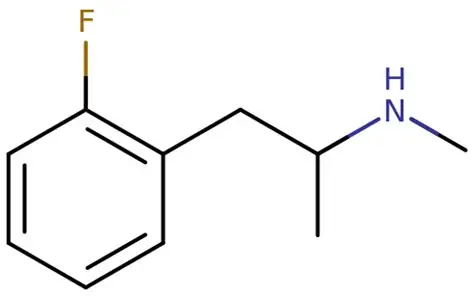
Section 2: Buy 2-FMA (2-Fluoromethamphetamine)
What is 2-FMA?
2-FMA (2-Fluoromethamphetamine) is a fluorinated amphetamine derivative and a ring-substituted analogue of methamphetamine. It belongs to the substituted amphetamine class.
Chemical Identity
- IUPAC Name: 1-(2-fluorophenyl)-N-methylpropan-2-amine
- Molecular Formula: C10H14FN
- Molecular Weight: 167.22 g/mol
- Appearance: White to off-white crystalline powder
Mechanism of Action
2-FMA acts primarily as a dopamine and norepinephrine reuptake inhibitor with weak serotonin activity:
| Transporter | Activity | IC50 (approx.) |
|---|---|---|
| DAT (Dopamine) | Moderate inhibition | 50-100 nM |
| NET (Norepinephrine) | Moderate inhibition | 50-100 nM |
| SERT (Serotonin) | Weak inhibition | >10,000 nM |
Key Research Findings
- Duration of action: 6-8 hours (longer than most amphetamines)
- Selectivity: DAT/NET selective with minimal SERT activity
- Research applications: Attention deficit studies, monoamine transporter selectivity research
Storage & Handling
- Conditions: Cool, dry, dark (15-25°C)
- Shelf life: 24 months
*For DAT-selective stimulant research, Buy 2 FMA as a certified reference material from Research Trade Lab Ltd.
Section 3: Buy 2-FEA (2-Fluoroethamphetamine)
What is 2-FEA?
2-FEA (2-Fluoroethamphetamine) is a fluorinated amphetamine derivative featuring an ethyl group on the amine nitrogen, distinguishing it from 2-FMA (which has a methyl group).
Chemical Identity
- IUPAC Name: 1-(2-fluorophenyl)-N-ethylpropan-2-amine
- Molecular Formula: C11H16FN
- Molecular Weight: 181.25 g/mol
- Key difference from 2-FMA: Ethyl group instead of methyl
Mechanism of Action
2-FEA exhibits a norepinephrine-predominant profile:
| Transporter | Activity Level |
|---|---|
| NET (Norepinephrine) | High (primary) |
| DAT (Dopamine) | Moderate |
| SERT (Serotonin) | Low |
Key Research Findings
- Duration: 4-6 hours
- Comparison to 2-FMA: Less dopaminergic, more noradrenergic
- Research applications: Norepinephrine transporter studies, SAR of N-alkyl substitutions
*For NET-predominant stimulant research, Buy 2-FEA from Research Trade Lab Ltd.*
Section 4: Buy 2-FPM (2-Fluorophenmetrazine)
What is 2-FPM?
2-FPM (2-Fluorophenmetrazine) is a fluorinated analogue of the stimulant phenmetrazine. Unlike amphetamines, phenmetrazines contain a morpholine ring structure.
Chemical Identity
- IUPAC Name: 2-(2-fluorophenyl)-3-methylmorpholine
- Molecular Formula: C11H14FNO
- Molecular Weight: 195.23 g/mol
- Class: Phenylmorpholine (phenmetrazine analogue)
Mechanism of Action
2-FPM acts as a dual dopamine and norepinephrine reuptake inhibitor:
- Primary mechanism: Reuptake inhibition (not release)
- Selectivity: DAT > NET >> SERT
- Distinction from amphetamines: Does not cause monoamine release
Key Research Findings
- Duration: 4-6 hours
- Potency: More potent than phenmetrazine
- Research applications: Reuptake inhibition mechanism studies, phenmetrazine SAR
*For phenmetrazine analogue research, Buy 2-FPM from Research Trade Lab Ltd.*
Section 5: Buy 2/3-FEA (Positional Isomer Blend)
What is 2/3-FEA?
2/3-FEA is a blended research formulation containing both 2-FEA and 3-FEA in a specific ratio. This blend allows researchers to study the combined effects of positional isomers.
Composition
- Component 1: 2-FEA (2-fluoroethamphetamine) – NET-predominant
- Component 2: 3-FEA (3-fluoroethamphetamine) – SERT-predominant
Research Applications
- Positional isomer synergy studies
- Mixed monoamine transporter modulation
- Comparative pharmacology of 2-position vs 3-position fluorination
*For positional isomer blend research, Buy 2/3-FEA from Research Trade Lab Ltd.*
Section 6: Buy 3-FA (3-Fluoroamphetamine)
What is 3-FA?
3-FA (3-Fluoroamphetamine) is a fluorinated amphetamine derivative with the fluorine atom at the 3-position of the phenyl ring.
Chemical Identity
- IUPAC Name: 1-(3-fluorophenyl)propan-2-amine
- Molecular Formula: C9H12FN
- Molecular Weight: 153.20 g/mol
- Key feature: 3-position fluorination
Mechanism of Action
3-FA exhibits a balanced monoamine profile:
| Transporter | Activity |
|---|---|
| DAT (Dopamine) | Moderate-High |
| NET (Norepinephrine) | Moderate-High |
| SERT (Serotonin) | Low-Moderate |
Key Research Findings
- Duration: 4-5 hours
- Comparison to 2-FMA: Less selective, more balanced
- Research applications: Positional isomer comparison studies
For balanced stimulant pharmacology research, Buy 3 FA from Research Trade Lab Ltd.
Section 7: Buy 3-FPM (3-Fluorophenmetrazine)
What is 3-FPM?
3-FPM (3-Fluorophenmetrazine) is a fluorinated phenmetrazine analogue with the fluorine atom at the 3-position. It is a positional isomer of 2-FPM.
Chemical Identity
- IUPAC Name: 2-(3-fluorophenyl)-3-methylmorpholine
- Molecular Formula: C11H14FNO
- Molecular Weight: 195.23 g/mol
- Class: Phenylmorpholine
Mechanism of Action
3-FPM acts as a potent dual reuptake inhibitor:
- Primary mechanism: DAT and NET reuptake inhibition
- Selectivity: DAT/NET selective
- Key feature: More potent than 2-FPM in some assays
Key Research Findings
- Duration: 5-7 hours
- Potency: High
- Research applications: Phenmetrazine SAR, reuptake inhibition kinetics
*For high-potency phenmetrazine research, Buy 3 FPM from Research Trade Lab Ltd.*
Section 8: Buy 3-MMA (3-Methoxymethamphetamine)
What is 3-MMA?
3-MMA (3-Methoxymethamphetamine) is a methamphetamine analogue with a methoxy group (-OCH3) at the 3-position, rather than a fluorine atom.
Chemical Identity
- IUPAC Name: 1-(3-methoxyphenyl)-N-methylpropan-2-amine
- Molecular Formula: C11H17NO
- Molecular Weight: 179.26 g/mol
- Key distinction: Methoxy group instead of fluoro
Mechanism of Action
3-MMA acts as a monoamine releaser (distinct from reuptake inhibitors):
- Primary mechanism: Promotes release of dopamine and norepinephrine
- Comparison: More similar to methamphetamine than to 2-FMA
Key Research Findings
- Duration: 3-5 hours
- Mechanism: Releaser (vs reuptake inhibitor)
- Research applications: Methamphetamine analogue SAR, releaser mechanism studies
For methamphetamine analogue research, Buy 3 MMA from Research Trade Lab Ltd.
Section 9: Buy 3-FEA (3-Fluoroethamphetamine)
What is 3-FEA?
3-FEA (3-Fluoroethamphetamine) is a fluorinated amphetamine derivative with the fluorine at the 3-position and an ethyl group on the amine nitrogen.
Chemical Identity
- IUPAC Name: 1-(3-fluorophenyl)-N-ethylpropan-2-amine
- Molecular Formula: C11H16FN
- Molecular Weight: 181.25 g/mol
- Positional isomer of: 2-FEA
Mechanism of Action
3-FEA exhibits a serotonin-predominant profile – unique among amphetamines:
| Transporter | Activity Level |
|---|---|
| SERT (Serotonin) | High (primary) |
| NET (Norepinephrine) | Moderate |
| DAT (Dopamine) | Low-Moderate |
Key Research Findings
- Duration: 5-7 hours
- Unique feature: Serotonergic effects distinguish it from other fluorinated amphetamines
- Research applications: Serotonin transporter studies, 3-position fluorination effects
*For serotonergic stimulant research, Buy 3-FEA from Research Trade Lab Ltd.*
Section 10: Buy 4-FMA (4-Fluoromethamphetamine)
What is 4-FMA?
4-FMA (4-Fluoromethamphetamine) is a fluorinated amphetamine derivative with the fluorine atom at the 4-position of the phenyl ring.
Chemical Identity
- IUPAC Name: 1-(4-fluorophenyl)-N-methylpropan-2-amine
- Molecular Formula: C10H14FN
- Molecular Weight: 167.22 g/mol
- Positional isomer of: 2-FMA
Mechanism of Action
4-FMA exhibits a unique profile including 5-HT2B receptor agonism:
| Target | Activity |
|---|---|
| DAT (Dopamine) | Moderate |
| NET (Norepinephrine) | Moderate |
| SERT (Serotonin) | Moderate |
| 5-HT2B Receptor | Agonist (unique to 4-position) |
Key Research Findings
- Duration: 5-6 hours
- Unique feature: 5-HT2B agonist activity (not seen with 2- or 3-position fluorination)
- Research applications: 5-HT2B receptor studies, position-dependent SAR
*For 5-HT2B and serotonergic stimulant research, Buy 4-FMA from Research Trade Lab Ltd.*
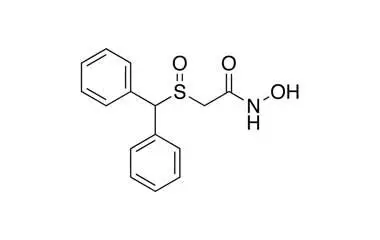
Section 11: Buy 4F-MPH (4-Fluoromethylphenidate)
What is 4F-MPH?
4F-MPH (4-Fluoromethylphenidate) is a fluorinated analogue of methylphenidate (Ritalin). It belongs to the phenidate class, not the amphetamine class.
Chemical Identity
- IUPAC Name: Methyl 2-(4-fluorophenyl)-2-(piperidin-2-yl)acetate
- Molecular Formula: C14H18FNO2
- Molecular Weight: 251.30 g/mol
- Class: Phenidate (piperidine derivative)
Mechanism of Action – Potent DAT Inhibitor
4F-MPH acts as a potent dopamine and norepinephrine reuptake inhibitor with high DAT selectivity:
| Parameter | Value |
|---|---|
| DAT (Dopamine) IC50 | ~61 nM (threo isomer) |
| NET (Norepinephrine) IC50 | ~31 nM (threo isomer) |
| SERT (Serotonin) IC50 | >100,000 nM |
| Potency vs Methylphenidate | ~3-5x more potent |
Key Research Findings
- Stereoisomer activity: Only the threo diastereomer is active; erythro isomer is inactive
- Duration: 6-8 hours
- Selectivity: Highly selective for DAT/NET over SERT
- Research applications: DAT inhibitor SAR, phenidate pharmacology
Storage & Handling
- Conditions: Cool, dry, dark
- Shelf life: 24 months
*For high-potency DAT inhibitor research, Buy 4F-MPH from Research Trade Lab Ltd.*
Section 12: Buy Blue Mandala Mix
What is Blue Mandala Mix?
Blue Mandala Mix is a research formulation blend containing multiple fluorinated stimulants. It is designed for analytical and comparative pharmacology studies.
Typical Composition (Research Grade)
While exact formulations vary by batch, Blue Mandala Mix generally contains:
- Fluorinated amphetamines (various positional isomers)
- Phenmetrazine analogues (e.g., 2-FPM, 3-FPM)
- Proprietary ratio of DAT/NET/SERT modulators
Research Applications
- Multi-target stimulant pharmacology
- Blended formulation analysis (HPLC, GC-MS)
- Comparative transporter studies
- Synergy and antagonism research
Analytical Considerations
- Expected purity: ≥98% total actives
- Individual component ratios: Vary; COA required for precise composition
- Recommended testing: HPLC-MS for component identification
For multi-component stimulant formulation research, Buy Blue Mandala Mix from Research Trade Lab Ltd.
Section 13: Positional Isomerism & SAR Comparison
The Importance of Fluorine Position
The position of the fluorine atom dramatically alters pharmacology:
| Position | Typical Profile | Example Compound |
|---|---|---|
| 2-position | DAT/NET selective, longer duration | 2-FMA, 2-FEA, 2-FPM |
| 3-position | Balanced or SERT-predominant | 3-FA, 3-FEA, 3-FPM |
| 4-position | 5-HT2B agonist, mixed profile | 4-FMA |
Comparative Potency Table
| Compound | Class | Primary Mechanism | Duration | Unique Feature |
|---|---|---|---|---|
| 2-FMA | Amphetamine | DAT/NET reuptake inhibition | 6-8 hr | Long duration |
| 2-FEA | Amphetamine | NET-predominant | 4-6 hr | N-ethyl group |
| 2-FPM | Phenmetrazine | DAT/NET reuptake | 4-6 hr | Morpholine ring |
| 2/3-FEA | Blend | Mixed isomers | Variable | Positional blend |
| 3-FA | Amphetamine | Balanced DAT/NET | 4-5 hr | 3-position |
| 3-FPM | Phenmetrazine | Potent DAT/NET | 5-7 hr | High potency |
| 3-MMA | Methamphetamine | Monoamine releaser | 3-5 hr | Methoxy group |
| 3-FEA | Amphetamine | SERT-predominant | 5-7 hr | Serotonergic |
| 4-FMA | Amphetamine | Mixed + 5-HT2B | 5-6 hr | 5-HT2B agonist |
| 4F-MPH | Phenidate | DAT/NET selective | 6-8 hr | Most potent DAT inhibitor |
| Blue Mandala | Blend | Multi-target | Variable | Complex formulation |
Research Implications
- 2-position compounds are ideal for DAT/NET-selective studies
- 3-position compounds offer serotonergic or balanced profiles
- 4-position compound (4-FMA) provides unique 5-HT2B data
- 4F-MPH is the most potent DAT inhibitor in this list
- Blends allow multi-target research in a single preparation
Section 14: Quality & Purity for Researchers – Buyer’s Guide
Critical Quality Metrics
| Parameter | Acceptable Range | Ideal Standard |
|---|---|---|
| Purity (HPLC) | ≥98% | ≥99.5% |
| Single largest impurity | <0.5% | <0.1% |
| Residual solvents | <5000 ppm | <1000 ppm |
| Heavy metals | <20 ppm | <5 ppm |
| Water content | <2% | <0.5% |
Analytical Documentation Required
- Certificate of Analysis (COA) – HPLC chromatogram
- Mass Spectrometry (MS) – Molecular weight confirmation
- NMR Spectroscopy – Structural verification
- Chirality analysis – For 4F-MPH (threo/erythro ratio)
Storage Recommendations
| Compound | Storage | Shelf Life |
|---|---|---|
| 2-FMA, 2-FEA, 3-FA, 3-FEA, 4-FMA | Cool, dry, dark | 24 months |
| 2-FPM, 3-FPM | Cool, dry, desiccated | 24 months |
| 3-MMA | Cool, dry, dark | 18 months |
| 4F-MPH | Cool, dry (threo isomer stable) | 24 months |
| Blue Mandala Mix | Cool, dry, dark | 12 months (blend stability) |
My Every Daily Blogs recommends Research Trade Lab Ltd for all eleven research compounds. They provide rigorous third-party testing and full COA documentation.
Section 15: Frequently Asked Questions
Q1: What is the difference between 2-FMA and 4F-MPH?
A: 2-FMA is a fluorinated amphetamine (DAT/NET reuptake inhibitor). 4F-MPH is a fluorinated phenidate (more potent DAT inhibitor, different chemical class).
Q2: How does 3-FEA differ from 2-FEA?
A: Positional isomerism: 3-FEA (3-position) is SERT-predominant; 2-FEA (2-position) is NET-predominant.
Q3: What is Blue Mandala Mix?
A: A research formulation blend containing multiple fluorinated stimulants for multi-target pharmacology studies.
Q4: Are these compounds legal to purchase for research?
A: Legal status varies by country. Many are unscheduled for laboratory research. Always verify local regulations.
Q5: Which compound is most selective for DAT?
A: 4F-MPH has the highest DAT selectivity (IC50 ~61 nM for threo isomer).
Q6: What is the significance of the threo isomer in 4F-MPH?
A: Only the threo diastereomer is pharmacologically active. The erythro isomer shows negligible DAT inhibition.
Q7: How should I store these compounds?
A: Cool, dry, dark conditions. Blue Mandala Mix has shorter shelf life (12 months) due to blend complexity.
Q8: Can these compounds be used in combination research?
A: Yes, but careful analytical verification is required. Blue Mandala Mix is specifically designed for multi-component studies.
Section 16: Conclusion & Future Research Directions
This overview of eleven fluorinated stimulants demonstrates the diversity of pharmacological profiles achievable through positional isomerism and structural modification:
- 2-position fluorination (2-FMA, 2-FEA, 2-FPM) → DAT/NET selective
- 3-position fluorination (3-FA, 3-FEA, 3-FPM) → Balanced or SERT-predominant
- 4-position fluorination (4-FMA, 4F-MPH) → Unique profiles including 5-HT2B agonism
- Methoxy substitution (3-MMA) → Releaser mechanism
- Blended formulations (Blue Mandala Mix, 2/3-FEA) → Multi-target research
Future Research Directions
- Isomer-specific kinetics – Detailed DAT/NET/SERT binding studies
- Metabolite identification – LC-MS/MS profiling for all compounds
- Chirality studies – For phenidate analogues
- Blend standardization – For Blue Mandala Mix
- Long-term stability – Under various storage conditions
Disclaimer: The content on My Every Daily Blogs is for informational purposes only. We do not condone the misuse of research chemicals. Our content is intended for licensed researchers and industrial professionals only.
Source: My Every Daily Blogs
Partner Link: Research Trade Lab Ltd